Germany is globally recognized for its advanced healthcare infrastructure, strong biomedical research environment, and strict regulatory oversight. The country maintains rigorous standards for clinical safety, ethical medical practice, and scientific validation. As regenerative medicine continues to evolve worldwide, Stem Cell Therapy in Germany remains an area of ongoing scientific interest for patients managing chronic and degenerative diseases. Alongside domestic healthcare pathways, some patients also explore structured international programs such as those offered by Stemcellcure.in, which operate under defined safety protocols and eligibility screening systems.
Stem cell therapy is grounded in biological science. Stem cells have the unique ability to self-renew and differentiate into specialized cell types when exposed to controlled laboratory and clinical conditions. Under appropriate medical supervision, these cells may assist in supporting tissue repair, regulating inflammatory responses, and enhancing cellular communication. However, it is important to emphasize that stem cell therapy is not a universal cure. It is considered a supportive medical approach that must be evaluated through evidence-based screening and individualized assessment.
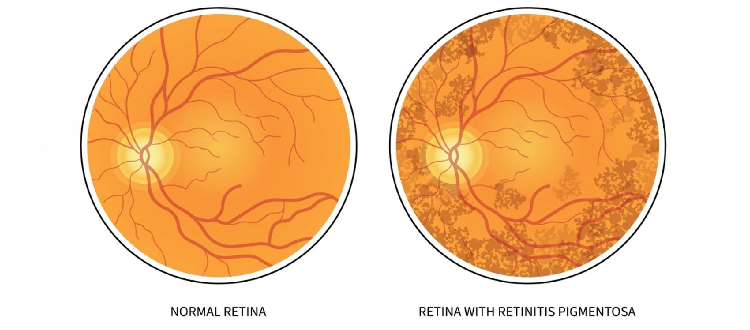
Patients researching Stem Cell Therapy in Germany commonly seek information for conditions such as:
• Osteoarthritis and joint degeneration
• Neurological disorders
• Autoimmune diseases
• Chronic inflammatory conditions
• Degenerative musculoskeletal problems
Germany’s regulatory environment prioritizes patient safety and ethical standards. Similarly, reputable international regenerative centers such as Stemcellcure.in follow structured medical frameworks that include:
• Ethical sourcing and documented procurement of stem cells
• Laboratory processing in certified sterile environments
• Administration under qualified medical supervision
• Comprehensive eligibility screening
• Transparent informed consent procedures
Before undergoing regenerative therapy, patients must complete a detailed medical evaluation. This typically includes diagnostic imaging (MRI, CT scans, X-rays when required), laboratory investigations, previous treatment history review, and overall health assessment. Not all patients qualify for stem cell therapy. Responsible providers recommend treatment only when clinically appropriate and supported by medical findings.
For individuals traveling from Germany for regenerative care, continuity of medical documentation is critical. Reliable centers ensure that patient records are reviewed thoroughly before treatment planning begins. After therapy, structured follow-up consultations—often conducted via telemedicine—support ongoing monitoring and medical communication once patients return home. This approach aligns with Germany’s emphasis on long-term healthcare accountability.
Scientific transparency is essential in maintaining trust. Outcomes of Stem Cell Therapy in Germany or abroad may vary depending on disease severity, stage of progression, immune response, age, and individual biological factors. Ethical providers clearly explain potential benefits, limitations, and possible risks without exaggeration. This responsible communication reflects Germany’s culture of evidence-based healthcare and informed patient decision-making.
Globally, regenerative medicine continues to advance through controlled clinical trials and peer-reviewed research. While promising, stem cell therapy remains under active scientific evaluation. Patients are encouraged to verify medical credentials, review available evidence, and avoid clinics making unrealistic claims.
Stemcellcure.in emphasizes patient safety, scientific integrity, and structured treatment planning. For individuals connected to Germany exploring regenerative medicine, careful research, professional consultation, and realistic expectations remain the foundation of responsible healthcare decisions.